sgoertzen
Members
-
Joined
-
Last visited
-
Country
United States
Everything posted by sgoertzen
-
Wellsky Transfusion (HCLL) Emergency Issue Module
WellSky has made some dramatic changes in their latest version to their Emergency Issue module. What version are you currently on?
-
Method Validation for Immucor Echo
Immucor (Werfen) supplies you with all the validation documents that you need in a nice binder. You just need to fill out all of the forms and attach the print-outs.
-
Correlation Testing in Blood Bank
Here is the form that we use. You need to have something written in policy that accounts for the expected variability of reactions when comparing different methods. We have multiple methods for ABO/Rh, Antibody Screen, Antigen Typing, Antibody ID, and AHG Crossmatch, so we have to do method comparison on all of these. TQ-0530F03 Method Comparison__blank_copy_id_10835032.pdf
-
"Keep Ahead" Orders
We used to have a "Keep Blood on Hand" (i.e. Keep Ahead) with our old computer system, but we discontinued that when we moved to Epic. I think it was a hold-over from "way back when" before electronic crossmatching. It was time for it to go!
-
Blood used organ donation services
At my institution. the Donor Network is now asking for 4-6 units of RBCs for organ perfusion for their machine after the organ has been harvested (similar to ECMO for the organ). Those RBC units will not ever touch the organ donor patient. Our policy is to always issue them the oldest O Pos units (uncrossmatched) we have on our shelf. They will rinse all of this banked blood out of the organ before transplanting it into the recipient, and it is added to the perfusate solution to provide oxygenation to the organ during transport from the donor hospital to the recipient hospital. AABB offered at least 1 very informative session at their annual meeting on this last year in Nashville, and I'm guessing that they will offer more in Houston this year (or perhaps an eCast session or articles in AABB News or Transfusion) since this practice is becoming more and more widespread. The unique nature of the process is proving to be a challenge for hospital transfusion services as far as who places the order, what testing is needed (if any), tracking for final disposition, what kind of records need to be kept because they are not being "transfused", billing of the products, etc..
-
Trauma Alert Procedures?
We ended up getting an undercounter Helmer fridge to hold 2 O Neg RBC units. This fridge is locked and hooked up to the Pyxis in the Trauma Bay in the ED. They must use the Pyxis to open the fridge and access those RBC units which triggers an alert to our Trauma pager up in the blood bank. This works well and has saved us so much time and resources because we no longer have to pack up units in a cooler and run them down to the ED on all level 1 traumas. It's well worth the cost of the fridge.
-
AABB 1.4 and 1.4.1 Risk Assessment
I would recommend adding "Risk Assessment" and "Risk Mitigation" as new sections in your Variance tracking system that must be completed by the reviewer for each variance documented. You should also add it to any of your internal audit report forms, equipment corrective action forms, and proficiency testing result review reports.
-
MaxQ MTP Coolers 3.0
I would be happy to share our validation plan for this cooler. Please send me a private message with your email.
-
Cold auto antibodies
This is also our policy. If the cold antibody is so strong (3+ to 4+) that we must use pre-warm tube testing in order to do the workup, we advise them to use a blood warmer regardless of the location where the transfusion is being given.
-
Psoralen Treated Platelet in Wellsky
We have no problem with this. I will connect you with our WellSky expert who built the system to work beautifully for us. Her name is Jill and her email is JHShaw@valleychildrens.org Please reach out to her and she can walk you through exactly how to do this. I will alert her to be expecting an email from you!
-
Digitizing Antibody Workup Files
We have WellSky and Epic and we scan everything and attach it to the patient in WellSky so it becomes a part of the patient's viewable blood bank testing history.
-
New PPID Rule Scripts
If you are using Epic, I can share the contact information with you to the people at my facility that set this up for us. Our Epic/Beaker system checks for appropriate PPID scanning at the time the specimen is received into the lab. If it fails, the sample is rejected unless it has the date/time and 2 sets of employee IDs written on the tube, but if the patient has no ABO/Rh history on file, then a 2nd specimen is also required.
-
Standard method for isoheme titers?
I've attached copies of our procedure and our worksheet. Our Heme/Onc docs also order them on our patients post-transplant, and we occasionally get them ordered on kids where they suspect some sort of immune deficiency disease. TO-310 Isohemagglutinin Workup - Test and Titer__uncontrolled_copy (2).pdf TO-310F01 Isohemagglutinin Test and Titer Worksheet__blank_copy_id_8428444.pdf
-
PEDIATRIC MASSIVE TRANSFUSION PROTOCOL
I've attached our MTP procedure and worksheet. We are a children's hospital with a level 2 pediatric trauma center.Massive Transfusion Protocol - MTP.pdfMassive Transfusion Protocol - MTP Worksheet.docx.pdf
-
Beaker Result Entry
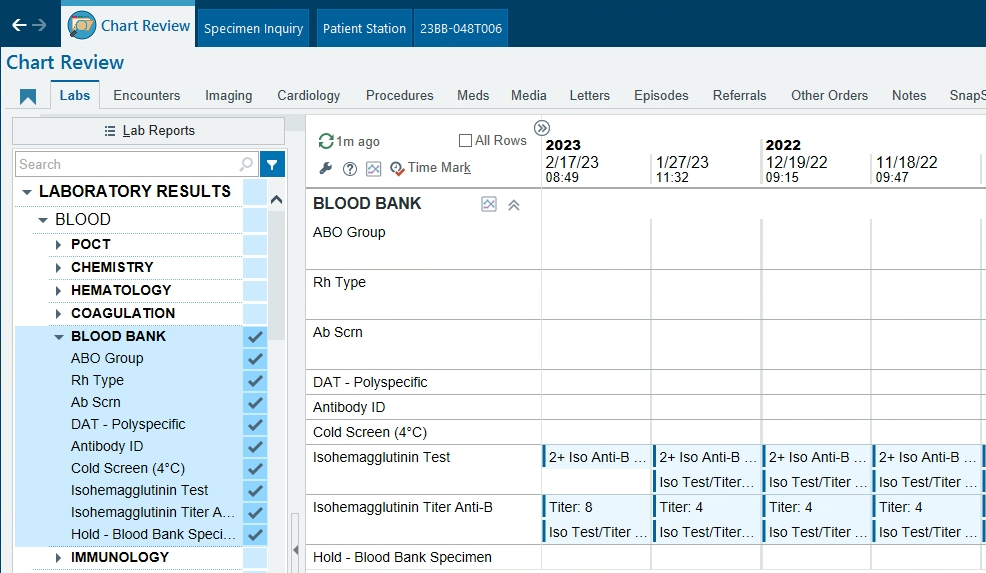
I work at a children's hospital and we use WellSky and Epic/Beaker. We built an orderable test called "Isohemagglutinin Test and Titer" specifically for reporting out the presence and strength of Anti-A and Anti-B. This test is not affiliated in any way with the regular patient Blood Type test. It's mainly ordered at my facility to monitor and follow patients with immune deficiencies or who have had an ABO mis-matched bone marrow or stem cell transplant, but it sounds like it would also meet your needs with heart transplants. I've attached our procedure and worksheet. This is how the results display in Epic. If you're interested in building something like this, I can put you in contact with our WellSky & Beaker IT gurus who built this for us. My contact is sgoertzen@valleychildrens.org TO-310 Isohemagglutinin Workup - Test and Titer__uncontrolled_copy (1).pdf TO-310F01 Isohemagglutinin Test and Titer Worksheet__blank_copy_id_7905995.pdf
-
RBC Transfusion thresholds for pediatrics
I'm the supervisor at a children's hospital in Central California and here are our indications for the transfusion of RBCs: Neonates: Term and near term neonates and infants < 4 months of age* Hgb/Hct < 7g/dl / 21% Stable anemia with no clinical manifestations Hgb/Hct < 10 g/dl / 30% Moderate cardiopulmonary disease Major surgery Increased oxygen (FiO2) requirement <35%, on CPAP lower setting Significant apnea or bradycardia, tachycardia or tachypnea Low weight gain Hgb/Hct <12 g/dl / 35% Fi02 requirement greater than 35%, on CPAP higher setting Recovering from major surgery Severe traumatic brain injury Significant deterioration of cardiorespiratory status Hgb/Hct < 15 g/dl / 45% FiO2 requirement > 35% Severe cardiopulmonary disease or congenital heart disease On extracorporeal membrane oxygenation (ECMO) *No clear transfusion RBC threshold guideline for low birth weight neonates (BW <1500gm) is available. Randomized clinical trial (Transfusion of Prematures) was started in 2013 and is ongoing. Pediatric patients >4 months old through adult Not bleeding Reasonable in almost all patients if Hgb/Hct < 7 g/dl / 21% Almost never indicated if Hgb/Hct >10 g/dl / 30% unless patient is on ECLS For Hgb between 7-10 g/dl (Hct between 21-30 %): Based on organ dysfunction and ability to handle inadequate oxygenation Respiratory or cardiac failure Chronic disorders of red cell production, severe platelet dysfunction Oncology patients Intra/perioperative conditions or significant bleeding Rapid blood loss exceeding >15% blood volume Intraoperative period as clinically determined by anesthesiology and/or surgeon Immediate postoperative period to restore hemodynamic stability We have built an alert in Epic with our "Prepare RBC" orders (both in mL and in Units) that warns the provider whenever they are placing an RBC order on a patient with a most recent Hgb value > 7 g/dl (or there is no recent Hgb value in the computer on that patient). This alert must be overridden with a reason from this drop down menu (below) in order for the provider to continue placing the order. We can run a report on all transfusions that triggered an Override when the order was placed (that also lists out the trigger value, the override reason, and the patient's problem list) and then the medical director performs an appropriateness review on only those outliers. BPA Overrides: RBC Orders (in mL) and (in Units): Warning if: No Hgb result or Most recent Hgb > 7 g/dl Appropriate criteria: Neonate w/Cardiopulmonary Disease Respiratory or Cardiac Failure ECLS Patient Sickle Cell Patient Thalassemia Patient Active Chemotherapy/Immunosuppressed Patient Hematopoietic Disorder Rapid Blood Loss HOLD for Pre-Op/Procedure Post-Op Hemodynamic Instability Other – specify as Comment
- Antibody Titer result form.
-
Labels
We created this label (Shamrock): It's actually a brilliant orange color. Perhaps Shamrock would let you purchase this one that we already created for ourselves (Valley Children's Hospital)?
-
Blood Bank Armbands
Re: We still have concerns about pre-op patients who aren't wearing any Epic band to scan when their pre-admit specimen is drawn. (I'm taking advice on how others manage these.) Likewise for outpatient transfusions. Epic told us that their system is not designed to use the process of banding outpatients and pre-op patients. WE INSISTED since 1) we've always banded any patient getting their blood drawn... especially for blood bank testing, 2) we were determined to meet AABB Std. 5.14.5.3) requiring an electronic (scanned) identification system, and 3) we decided that we were NOT going to go backwards after all these years and create a new system in Epic that was less safe just because they said that's their design. I insisted that PPID scanning be used for the specimen collection/labeling and that the same armband be presented on the day of their admission or outpatient transfusion. The patient is given strict instructions (an instruction sheet that they must sign and is scanned into the EMR) that they are to keep the band on or at least have it in their possession on the day of admission/transfusion. The original band used for specimen collection is replaced with their new encounter band only after the 2 bands are compared side-by-side and match exactly for Name, MRN, DOB. It was a bit of a struggle to get everyone on board to veer from the Epic "Foundation" methods, but we were finally able to convince people that this was a significant patient safety issue and was necessary.
-
Blood Bank Armbands
I highly recommend you use this opportunity to discontinue using a separate BB ID band. Using the patient’s regular ID band works great. If you’re using scanned PPID from the wristband for specimen collection & labeling, adding another ID band into the process no longer adds any safety benefits. You’ve already created your closed loop system using the regular hospital ID band and scanning it for both specimen collection and blood administration in BPAM.
-
Procedure for Reconstituted Whole Blood
Here is our procedure. TO-480 Mixing Blood for Exchange Transfusions and CRRT.docx
-
Emergency Release Physician Signature in EPIC
When blood bank is called to release uncrossmatched blood, we specifically ask who the ordering physician is. The blood banker then places an order in EPIC called "Release of Uncrossmatched Blood" which is the statement (taken from AABB Standards) about the situation being critical enough to warrant the release of blood before compatibility testing is completed. This order must be electronically co-signed in EPIC by the physician that we name as the ordering physician. We have EPIC and WellSky (Mediware HCLL), but it shouldn't matter what blood bank computer system you have since this is all done (ordered/cosigned) in EPIC. I'm attaching our uncrossmatched worksheet that includes the instructions that we follow to place this order in EPIC and assign it to the ordering physician for co-sign. I check each day to make sure it gets co-signed. Once signed, I print the order with the co-sign tracking information, attach it to our worksheet, and file the paperwork for easy retrieval during future inspections/assessments. TO-381F01 Release of Uncrossmatched Blood.docx
-
Tube Antibody Titers: Yes or No to Enhancement?
We have a busy Fetal/Maternal Center for high risk pregnancies at our hospital. Our primary method of testing is solid phase (Capture) for antibody detection and identification, and our secondary method is PEG tube testing. The perinatologists are requesting antibody titers on all pregnant women with clinically significant alloantibodies. We have repeatedly seen clinically significant antibodies that react 3+ to 4+ with solid phase methodology that end up being "too weak to titer" when we move them to the tube for saline/37C/AHG titering. Even the non-diluted plasma reacts negatively in the tube at 37C and AHG without any enhancement. This is confusing to the perinatologists and I understand why. It doesn't make sense to them that a 4+ strong antibody can be too weak to titer. Does anyone else frequently experience this? I'm just curious whether anyone is routinely using any kind of enhancement when performing antibody titers on known clinically significant antibodies (such as CcDEe, K, Fy, Jk, etc). I know that the CAP ABT Survey choices for titer "diluent" are saline, 0.5% albumin, 6% albumin, 22% albumin and their "technique" choices include various versions of IS, RT, 37C, AHG, DTT (we're using saline w/ 37C incubation and poly AHG). With the exclusion of a gel titer, if you have a procedure (with criteria) for performing an "enhanced" titer, would you be willing to share it? For those of you using albumin for diluent, do you find this helps enhance the reaction of the antibody and what strength of albumin do you use?
-
Neonate Platelet Aliquots - references?
Hi Sonya - yes, it's a custom label we had them make for us at Shamrock. If you like it, they may just let you order ours rather than create your own! Hope you are all well and happy in San Diego!
-
Neonate Platelet Aliquots - references?
We apply this custom label to all PR platelets and PR platelet aliquots (see attached picture). We're a children's hospital and we've been giving PR platelets to neonates since March 2017 with no problems. PR Platelets.docx