Kelly Guenthner
Members
-
Joined
-
Last visited
-
Country
United States
Everything posted by Kelly Guenthner
-
Standard regarding sharing of pre-transfusion testing results between hospitals
Your policy (to draw and test your own specimen) is the strictest interpretation of the standards (and one that most blood banks likely follow), but I do believe you'll find systems that share MRNs, share LIS, share policy & procedure (may even share technical staff) that will use pretransfusion results performed in blood bank A to transfuse the patient at location B by technical staff in blood bank B and are doing so within AABB and CAP standards. All that is to say, I don't think you'll find a standard that explicitly says you have to draw and test your own specimen at your own physical location. We come to that conclusion because of all the other standards.
-
Standard regarding sharing of pre-transfusion testing results between hospitals
I think this is the key: Is initial testing at that ED is under one patient identifier/armband and the testing at your facility under a completely different patient identifier/armband? It's less about the testing, more about the patient identification. From CAP: TRM.40230 Specimen Labeling for Pretransfusion Testing Phase II All blood samples used for pretransfusion testing are labeled at the time of specimen collection in the presence of the patient with: 1. Patient's first and last name 2. Unique identification number 3. Date of collection 4. A method to identify the individual collecting the specimen. NOTE: Blood specimens collected for pretransfusion testing must be positively and completely identified and labeled before leaving the patient. Acceptable practices for positive identification of patient and blood specimen labels must be defined in the procedure manual and may include visual inspection and/or an electronic system to read the identifying information contained in bar codes or radio-frequency identification (RFID) microchips or the patient's wristband. Acceptable practices for generating specimen labels must be defined in the procedure manual (refer to GEN.40490) and may include electronic devices utilizing information encoded in bar codes or RFID microchips. There must be a dependable method to identify the individual who collected the blood specimen, such as initials or another identifier on the tube, or an electronic record. Evidence of Compliance ✓ Properly labeled blood specimens AND ✓ Records identifying the individual collecting pretransfusion testing specimens
-
FDA requirements for 2 people to issue blood products for transfusion.
We use a hands-off process where the floor faxes their request for blood, the blood banker sends the unit via the pneumatic tube and the floor faxes/sends back the signed copy of the unit tag. I am finding it difficult to come up with a routine transfusion scenario that does not involve 2 people at issue (one on the floor, one in the blood bank). I can come up with a couple of CAP requirements that you could cite, though they don't explicitly say 2 people at issue, you could certainly argue that your way of satisfying the requirement is to have one person from the floor, one person in the blood bank at issue.
-
ABID Using Mixed Methodologies?
We use Ortho Panel A (gel) as our primary method; Immucor/Werfen panocell as supplement (diluting to 0.8% and using gel; will also use PEG/LISS IAT as warranted). Additionally, we save a couple months worth of expired cells to use for selected cells, when our in-dated cells don't have the right antigen combo.
-
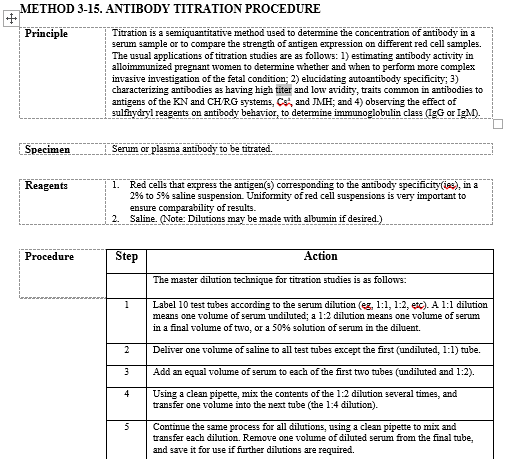
Titer controls
We also do titers on the Vision. We keep an aliquot of the previous titer, but only test it in parallel with the current if there is a significant difference to the current (titer jumps/drops 2 or more dilutions [The increase of 2 or more is our alert/critical value]). We titer against screen cells, which are QC'd daily and do not do any additional titer QC.
-
Antibody Titers Performed on Gel
-
Correlation Testing in Blood Bank
Our "methods" are automated gel, manual gel & tube (PEG). We do not evaluate our alternate enhancements as different "methods".
-
Computer Sign-In for Lab Trainees
Everybody gets a unique login, here (students and new employees) and only results under their own login. In blood bank, we go the extra step to put "reviewed by:___" comments on all of our students' work.
-
issue: Multiple Type and Screen ordered in Epic. Lab system is Soft Bank.
We function with your current workflow description and @jshepherd's work-around description.
-
"Keep Ahead" Orders
We allow keep ahead orders, but we do electronic crossmatch, upon issue. So a keep ahead order, for us, is just a peek at what's on the shelf and adjusting stock, as necessary. Additionally, hardly any of our providers use keep ahead (mostly just active cardiothoracic surgeries).
-
ABO Retypes
Our prenatal TS come from outside clinics and are registered with outside referral medical record numbers that are different from the hospital medical record numbers (under which they would be transfused). Because they are immunohematology and not pre-transfusion, we do not require a second ABORH. Prenatal referral records will be linked to hospital records, but the ABORH will not count as a second type on record in transfusion situations.
-
Thawed FFP
From the CAP Transfusion Medicine Checklist: COMPONENT PREPARATION, STORAGE AND MODIFICATION The following component definitions are offered as a convenience: Component Definition Fresh Frozen Plasma (FFP) Plasma frozen within 8 hours of collection after being separated from a unit of whole blood or frozen within 6 hours after collection by apheresis Plasma Frozen Within 24 Hours After Phlebotomy Plasma separated from whole blood and frozen between 8-24 hours after collection FFP, Thawed Fresh Frozen Plasma thawed between 30-37°C, then stored at 1-6°C for up to 24 hours Plasma Frozen Within 24 Hours After Phlebotomy, Thawed Plasma frozen within 24 hours of collection that has been thawed between 30-37°C, then stored at 1-6°C for up to 24 hours Thawed Plasma “FFP, Thawed” or “Plasma Frozen Within 24 hours After Phlebotomy, Thawed” which is stored in a closed system at 1-6°C for 1-5 days after thawing and the related standard: TRM.44525 Thawed Plasma Label Phase II If Fresh Frozen Plasma or plasma frozen within 24 hours of collection is thawed at 30 to 37°C and maintained at 1 to 6°C for one to five days, it is relabeled as "Thawed Plasma". My interpretation is only Thawed Plasma, (either FFP or FP24), stored >24 hours (1-5 days) requires relabeling.
-
HemeLabTalk: Iron
I just answered this question. My Score FAIL
-
HemeLabTalk: Oxygen transport
I just answered this question. My Score FAIL
-
Temp indicators for blood products
Not the original poster, but we use them in coolers to the OR and shipping boxes we send to outlying clients. An electronic data logger accompanies units in the cooler to ensure the cooler has remained at temp; Safe-T-Vue ensures that the unit has remained at temp. In our shipping boxes, clients have the option of using the Safe-T-Vue as the sole indicator; though most back it up with a temp gun/thermometer reading on arrival. We're going to stick with Safe-T-Vue, despite the price jump.
-
Ortho Gel combo workstation
We're still rockin' this original setup on our single combined Ortho Workstation. It's trash, but it works. If/when it's time to replace our 2 old separate setups, we will try to source an alternative.
-
MaxQ/Max+ cooler users...
That's the one! And the handle is integrated, but has 2 little screws that allow it to be removed the rest of the integrated assembly. Should be an easy replacement, once we find a match! (We're doing the legwork on this particular project, as our Biomed is very busy!)
-
MaxQ/Max+ cooler users...
Has anyone successfully replaced the telescoping luggage handles on their MaxQ coolers, either with manufacturer provided replacements or "after-market" parts (Amazon?)? We have been using (and loving) our MaxQ coolers for about 4.5 years now. so much so that we've banged up the telescoping luggage handles pretty good. When querying the manufacturer about replacing the handles, they say "Nope! Gotta replace the whole box!"
-
Pipettes (droppers)??
Not looking for glass, per se... but, we had still been using traditional soda lime blood bank pipettes You know... because... "That's what we always use!" . I guess no one here cares about safety! When we ran into manufacturing shortages of those, we used up the leftover glass borosilicate pipettes from the core lab. Now, we're on to plastic (Cardinal CH5214-12 or CHB521412), which is probably what we'll stick with. Thanks for your input, everyone!
-
Antibody identification art or science
I believe OP is referring to this publication: https://www.aabb.org/aabb-store/product/antibody-identification-art-or-science-a-case-study-approach---print-2285
-
Pipettes (droppers)??
Well, I feel like a real dope for asking, but, what pipettes are y'all using for your bench testing, these days? My purchasing coordinator is at her absolute wits-end trying to find blood bank suitable pipettes. She has orders in to every manufacturer imaginable for standard blood bank soda lime glass pipettes. Additionally, we've seemingly burned through every available stash of borosilicate glass pipettes she can get her hands on. We've now moved on to the smallest available plastic transfer pipettes (that still deliver a standard blood bank-sized drop), but may need to switch to larger transfer pipettes! Anyone have a source they want to share?
-
Preop Specimen
We do 21 days.
-
Helmer Freezer
We printed on cardstock, affixed with self-laminating sheets, and labeled with the freezer at ambient temperature.
-
BloodBankTalk: E-mail and panic values
I just answered this question. My Score PASS
-
BloodBankTalk: Clinical Aspects of Transfusion Reactions
I just answered this question. My Score FAIL