-
Intrauterine Transfusion Process
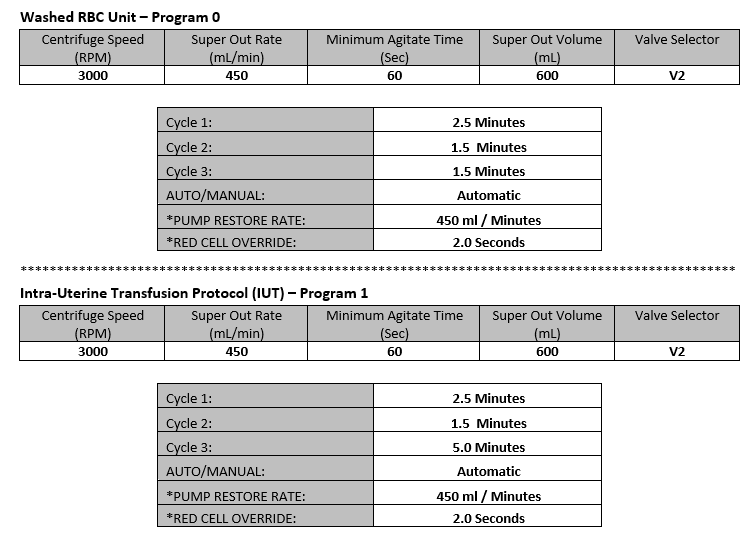
We are having an issue with our intrauterine wash procedure and the Cobe 2991. When we wash a unit for an IUT, we end up with at least 200mL of saline left over in the bag. We are using the same process for washing RBC units but have added a final 5 minute spin to pack the RBC in the IUT. Are settings are listed below. Has anyone had this problem or can shed some light on what we may be missing? Any help is appreciated.
-
Platelet Mapping and Rotems
We have 3 TEG 3000s in the blood bank. Our Core lab has just brought in 4 Rotems and we would desperately love to get the TEGs out of the blood banks. The problem is that the Rotems do not run platelet mappings and the TEG does. We were told that running the Rotem with the drug/aspirin cartridge on Verify Now would provide comparable result as the platelet mapping. Our Medical Director said the Rotems/Verify Now result does not provide results for platelet inhibition which he thinks is needed. Are there any location that use the Rotems/Verify Now for their patient and what is done for platelet inhibition? Any assistance is very appreciated.
-
SAbarry
For the facilities that perform 'in vivo' crossmatches, do you mind sharing your procedure? The procedure we use involves washing an antigen matching units for Rh, K, and Kidd. A pre-plasma hemoglobin is testing on the patient, followed the transfusion of 10mLs. A post transfusion hemoglobin is again collected and tested. Pre and post results are compared and acceptable RBC compatibility is determined. A change of less than or equal to 10mg/dl-unit will be transfused; 11-19 mg/dl physician must approve transfusion; greater than or equal to 20mg/dl do not transfuse unit, consult physician to determine if another unit should be prepared. This process is timely and creates delays in patient care. Are your procedure more streamlined? We us 'in vivo' at my current location but I have not used it anywhere in my 20+ years of being a blood bank.
-
SAbarry
Thanks for your replies. We very much want to get away from using the 'in vivo' crossmatch procedure. Unfortunately this procedure has been used in our facility for over 20 years and the physicians are reluctant to let it go. Malcolm, if crossmatched units are incompatible, do you ask for an emergency signature from the physician? How often is the WAA work-up repeated?
-
SAbarry
What do other do require for transfusing patient's with a strong WAA or strong CAA? What type of crossmatch is used and what antigen are being honored for these patients? We currently use 'in vivo' or biological crossmatches but am curious to see what others do. This is what we do for WAA: T&S-Initial testing will be on ECHO; If patient has a known WAA, testing is performed with LISS to avoid WAA interference. Perform panel and DAT. Expected results panagglutination and DAT positive. Perform elution studies. Expected result, panagglutination with eluate. Complete phenotype or partial phenotype (Rh, Kell, Kidd) if patient has not been transfused. Perform PeG auto or allo-adsorptions with W.A.R.M. treated cells as necessary. If patient has been transfused, perform triple allo-adsorption with donor cells. Send patient sample for molecular phenotyping for future transfusions. Crossmatch with neat plasma. If incompatible, perform ‘in vivo’ crossmatch with Rh, Kell, Kidd matched units with emergency release. CAA with incompatible blood are also 'in vivo' crossmatched with Rh, Kell, Kidd matched units with emergency release
-
How do you determine if ant-M is significant?
Yes I remembered that as well. The wording changed with the 16th Edition to only mention the 37C testing and stated that anti-M could be avoided with not performing immediate spin crossmatches and antibody screen testing. The wording was changed again with the 17th Edition and now only references 37C reactivity. The BBTS 28th Edition states that (paraphased) only a 37C incubation phase needs to be performed to determine clinical significance. I have written to AABB for clarification. Unfortunately the reply said I could expect an answer in about 4 weeks. Thanks to everyone for their responses. I will keep you posted on the AABB reply.
-
How do you determine if ant-M is significant?
What is everyones take on the following: How do you determine clinical significance of an anti-M? What I am consistently seeing and what we have all learned is that anti-M should be considered clinically significant when reactive at 37C and AHG phase to which I agree. Unfortunately with the introduction of automation (both gel and solid phase) the phase of testing is limited to a Coombs phase. The 17th Edition Technical Manual still references 37C testing but if tube or LISS testing is not being used in the hospital setting, is it acceptable to rely only on the AHG result provided by automation? e.g. Antibody screen is negative but immediate screen crossmatches are positive. Anti-M is identified in panel studies. I would select random units (not M antigen negative) and perform AHG crossmatches considering the anti-M is not significant since reactivity has not been detected at AHG. Some individuals are questioning the necessity of repeating the antibody screen in tubes to include the 37C phase. Do anyone know of a reference that states clinical significance can be identified by reactivity at 37C or AHG? I just want to prevent the additional extra work for my staff to prove the clinical insignificance of an antibody. Much frustration over anti-M so hopefully someone can help!
-
Heat Block Temperature Range
Most places I have worked have used a heat block range of 37C (+/- 2). My new place of work has a heat block range of 37C (+/- 1). I have not been able to find any manufacturing guidelines for the old Thermolynes. The current Technical Manual states that a +/- 1 is acceptable but it doesn't say that a +/- 2 is unacceptable. What are people using? I'd like to change our current range since I have discovered some QC that has been out of range and would like to prevent the corrective action investigation I now have to perform.